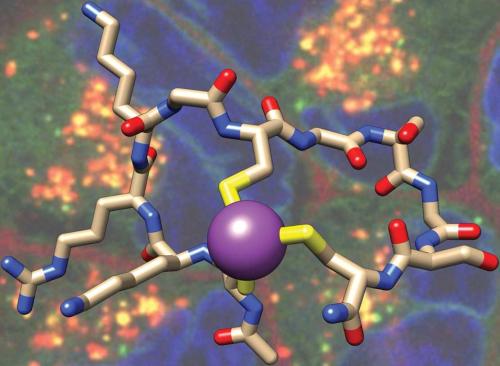
Cell-penetrating peptides play a crucial role in transporting cargo, including drugs, across human cell membranes. Enhancing the effectiveness of this process involves forcing the peptide into a more constrained shape, enabling sufficient cell penetration at lower concentrations. A straightforward method for constraining peptides into a bicyclic shape involves BiIII binding to three cysteine residues, a technique pioneered by the Nitsche group at the Australian National University (ANU). In a recent development, an international team comprising scientists from the ANU, Free University of Berlin (Germany), University of Sydney and Heidelberg University (Germany) has applied this strategy to explore peptide–bismuth bicycles as a new class of cell-penetrating peptides (Voss S., Adair L.D., Achazi K., Kim H., Bergemann S., Bartenschlager R., New E.J., Rademann J., Nitsche C. Angew. Chem. Int. Ed. 2024, 63, e202318615). These peptides outperform conventional cell-penetrating peptides such as TAT or octaarginine and require as little as three positive charges to traverse cell membranes at nanomolar concentrations. While these peptides can be linked to fluorescent dyes for monitoring cellular uptake through microscopy, uptake can alternatively be tracked by mass spectrometry of bismuth, providing a novel method to study cell-penetrating peptides without the need for fluorescent labels.


The Royal Australian Chemical Institute's magazine for and about the chemical science professions