Contrary to sensational media reports and class actions, global regulators consider glyphosate not to be carcinogenic. Consequences could be dire if market forces dictate its phase-out, says Ivan Kennedy.
The herbicide glyphosate was released by the Monsanto agrochemical company to the global market, with the trade name of Roundup, in 1974. Because of its unique properties, this new chemical met many of the requirements for an ideal product for controlling weeds, with minimal environmental and human impacts. Yet despite this wonderful beginning, a half century later glyphosate now finds itself accused in courts as harmful, the subject of global class actions.
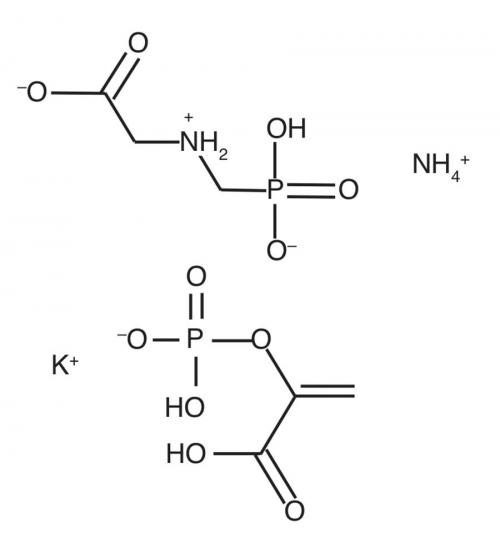
Glyphosate has a highly specific action in mimicking phosphoenolpyruvate (PEP), substrate to the plant enzyme EPSP synthase. EPSP synthase is essential for shikimate synthesis, a precursor for aromatic amino acids such as phenylalanine and tyrosine and for lignin needed for wood. This enzyme is not found in animals because aromatic amino acids are dietary, explaining this unique specificity of action.
Another advantage is glyphosate’s very low toxicity to animals, with LD50 values greater than those for common salt. (The LD50 value is the acute dose that will cause fatality in 50% of a population of test animals.) As a moderately water-soluble salt ionising from each of its carboxylic acid, amino and phosphate moieties, glyphosate is rapidly excreted in urine if ingested. As a zwitterion salt that dissociates, it has little or no volatility, even under acid conditions.
Glyphosate is quickly inactivated in soil from strong binding to its phosphate group, giving a residue that is degraded mainly by decarboxylation to aminomethylphosphonic acid (AMPA), which may be persistent when soil is dry, also bonded to soil by its phosphate group. If concentrated formulations are diluted with water containing soil, glyphosate is rendered inactive by this binding reaction.
Two aspects of its properties led to it becoming the world’s major marketed herbicide (Duke and Powles, doi.org/10.1002/ps.1518), almost one million tonnes annually in many different commercial formulations, since it is now out of patent.
First, it is a broad-spectrum herbicide, which is highly toxic to most annual plant species when applied to leaves but has no residual activity in soil; this inactivation makes it ideal for low tillage and no-till agriculture. This more sustainable agriculture has become very popular because it removes the need for costly ploughing of land in fallow before planting of crops. This has improved crop yields, reducing erosion and the cost of weed control. It also conserves water and nutrients in the soil profile and reduces the use of fossil fuels for ploughing with tillage implements, producing far less CO2 emissions.
Second, Monsanto’s success in producing genetically modified (GM) plants with closed EPSP synthases made it a favoured target for Roundup Ready crops designed to be herbicide resistant. Several crops have been provided with alternative EPSP genes from bacteria resistant to the herbicidal effect of glyphosate, such as soybeans, cotton, canola (rape) and maize.
Risk and regulation
Given these properties, it is not surprising that glyphosate is so highly favoured by world agriculture. No substitute technology is available that provides such a broad range of advantages. However, the many glyphosate-based formulations, but particularly Roundup, have been under attack by those opposed to GM technology, partly because of its use in Roundup Ready crops. Indeed, some unscientific papers published in poorly reviewed journals seemed to have had no other reason for their publication than to oppose its use. Such papers include speculation that glyphosate could be the cause of many unexplained diseases, but with no epidemiological evidence of an increase in occurrence as the use of glyphosate increases. One of my own students, fearing negative effects of glyphosate in the environment, found it did no harm when tested for effects on growth of beneficial environmental microbes such as Chlorella, and nitrogen-fixing Rhizobium and Anabaena. The effects were either null or actual stimulation of growth.
This is not to claim that glyphosate has no management problems. Increasing resistance to its action through genetic mutation of weeds is one issue, requiring that other more residual herbicides also be applied strategically. The search for other chemicals with a similar range of advantages is highly desirable but no ideal substitute for glyphosate is on the horizon (Beckie et al., doi.org/10.3390/plants9010096) and more toxic chemicals might be needed if glyphosate is not available. However, targeted spraying using digital technology might assist by reducing the intensity of applications.
Many chemical pesticides are hazardous and their use is legally regulated to maximise their effectiveness and safety. My experience is that the Australian Pesticides and Veterinary Medicines Authority (APVMA) is highly professional regarding its independence in risk management. Pesticides are usually applied in a mixture containing adjuvants such as emulsifiers or solvents to minimise the need for active ingredients. Individual toxicity to animals is indicated by the LD50. Conditions for use given on product labels aim to ensure that the probability of chronic exposure to the active ingredient is orders of magnitude less than such values, although accidental or deliberate ingestion of formulations can be dangerous and is the main cause of global mortality. Typically, laboratory rats or mice are used to establish inherent toxicity, including testing in vitro for mutagenicity and carcinogenicity. Tests required by regulators must be performed under conditions that mimic likely modes of exposure in humans, such as by ingestion, by respiration or through the skin. Establishing a legally approved label for a new product intended for global sales can cost hundreds of millions of dollars.
Pesticide effects on human health
To measure the effect of significant human exposure to any industrial chemicals, medical authorities conduct epidemiological surveys of human health to identify linked patterns or clusters of disease. Such surveys may rely on anecdotal recall of chemical usage from many years previously. Moreover, surveys are confounded by the multiplicity of chemicals used in particular industries. For example, farmers may have been exposed to many chemicals thought carcinogenic, including fuels containing benzene, cleaning solvents such as trichloroethylene, as well as several halogenated pesticides now discontinued.
Since 1974, a very large body of quality-controlled evidence on glyphosate has been critically reviewed by specialist scientists working for regulatory agencies in the European Union, the US, Canada, Japan, Australia, Brazil, and the Food and Agricultural Agency of WHO, as well as in the US Agricultural Health Study. None of these studies shows a significant causal link between glyphosate and cancer. In New South Wales, the incidence of all cancers (Depczynski et al., doi.org/10.1186/s12885-017-3912-2) in farm residents 45 years and older was lower than in urban residents, with no differences for non-Hodgkin lymphoma in farmers compared with urban residents.
These results are consistent with glyphosate’s benign structure consisting of connected groups of carboxylate, amino and phosphate. Its main degradation product in soil, AMPA, also a zwitterion and shown to be no more toxic than salt, is even simpler in structure. Its likely maximum concentration when bound in soil is about 50 times lower than the no-effect concentration on a range of soil animal species and microbes – a similar safety margin to that observed with glyphosate (von Mérey et al., doi.org/10.1002/etc.3438).
Glyphosate also has low immunogenicity – despite several attempts using glyphosate as a hapten linked to proteins injected into rabbits, we failed to raise sensitive antibodies. In glyphosate, the amino acid glycine is bonded to phosphate by a methylene bridge, yielding a molecule showing none of the characteristics usually associated with mutagenicity or carcinogenicity, such as free radical formation, aromaticity, heavy metal content or fat-soluble halogen content. Furthermore, no residues of glyphosate can accumulate in animals.
Although the International Agency for Research on Cancer (IARC) deemed glyphosate to be ‘probably carcinogenic to humans (Group 2A)’, this surprise decision gave no quantitative weight to risk of exposure and it may have been meant to encourage further research on genotoxicity. This approach contrasts starkly with Paracelsus’ dictum that ‘the dose is the poison’. With only a limited data set from scientific journals being considered, IARC claimed that there was strong evidence of genotoxicity and oxidative stress in tissue cultures at high concentrations (IARC Monograph 2015). This conclusion is strongly contested by much previous regulatory data and the ‘strong evidence of genotoxicity’ involved an incorrect interpretation of a key paper regarding micronuclei IARC cites (Bolognesi et al., doi.org/10.1080/15287390902929741; see critique in Solomon, doi.org/10.1564/v28_aug_08).
IARC’s hazard-based classification contrasts with the more stringent risk-based, weight-of-evidence approach used by all international regulatory agencies, including WHO (IARC’s parent agency), the European Food Safety Agency, Health Canada, US Environmental Protection Agency (USEPA), the APVMA (2017, see website) and several others. Perhaps influenced by the highly precautionary attitude in the European Union where the decision was made, IARC should have given a more guarded warning instead, such as ‘possibly carcinogenic’. The other internationally regarded authority for classifying carcinogens, the National Toxicology Program supported by the American Cancer Society, does not even list glyphosate as possibly a carcinogen.
In my opinion, IARC should reconsider its classification or reform its methods to be more transparently quantitative, using a risk approach that includes the likelihood of exposure. IARC claims it prefers to rely on peer-reviewed literature for its decisions, but this literature is of variable quality and the IARC’s working party failed to consider many quality-assured studies conducted for national regulators. These stringent requirements to meet regulatory guidelines are the main reason for the high costs (approximately US$250 million) of bringing any new chemical product to market. It will be difficult for the global agricultural industry to trust IARC in future without some reform and this threatens affordable food production globally.
In 2019, a Washington University meta-analysis (Zhang et al., doi.org/10.1016/j.mrrev.2019.02.001) suggesting that glyphosate increased the risk of non-Hodgkin lymphoma (NHL) by 41% was seized on by the media as a ‘compelling link’. The statistical result of their reanalysis selecting longer exposures was that NHL had an increased ‘meta relative risk’ (RR) of 1.41 from a previous RR of 1.23, less than RR 2.0 required for significance. Their use of a subset of applicators with longer exposure to glyphosate, who are more likely to have been exposed to other carcinogenic compounds, explains the slight increase in RR value. Their conclusion that their meta-analysis ‘suggests a compelling link between exposures to GBHs [glyphosate-based herbicides] and increased risk for NHL’ was only weakly supported by their study, bolstered with speculative effects regarding immunosuppression and endocrine disruption. Furthermore, the updated Agricultural Health Study of 54 251 applicators in the US (Andreotti et al., doi.org/10.1093/jnci/djx233) found no association between glyphosate and NHL, disproving their hypothesis of a 41% increase. If glyphosate does have a causal influence on lymphomas, one would also expect to see a dose response in test mice. Even in the small proportion of studies with mice and rats indicating a marginal statistical effect, no dose response has been observed.
Consequences of ‘knee-jerk’ bans
Research to provide new pesticides has many similarities to research in medicinal chemistry. Both require the screening of large numbers of possible candidate chemicals followed by confirmation of their effectiveness and freedom from unintended consequences. Only after years of such expensive trials can they be legally registered for use. World agriculture will resist attempts to restrict the use of glyphosate. However, market forces in trade may dictate that glyphosate will be phased out, whether by government decree or from commercial decisions. This would be a very frustrating outcome, given the huge intellectual and financial investment in this successful technology and the uncontrolled risk of negative consequences on human health from substitute herbicides.
So, what might be used instead? Substituting glyphosate with other less safe herbicides or even returning to mechanical control would be a huge backward step for productivity of farmers and safety in the environment. Substitutes will be more toxic and probably less effective, having to be used more often. Even if the precautionary principle is applied, it should retain the use of glyphosate because it is less harmful than any of the likely substitutes. Only when a substitute can be found that has similar advantages to glyphosate should glyphosate’s use be discontinued. In any case, its arguable overuse at present may be gradually reduced by new approaches to weeding, such as targeted digital technology available to modern agriculture.
Court actions
Unfortunately, IARC’s decision fostered a flurry of court actions in the US and elsewhere regarding Roundup, seeking damages from Monsanto and its current owner, the German company Bayer. These actions have focused on cases of non-Hodgkin lymphoma claimed to result from exposure to glyphosate. However, it is debatable whether juries have the capability to make objective decisions of such a technical nature, particularly when judges decline to allow evidence from USEPA experts. Jury decisions based at least partly on sympathy for such claimants are inevitable. The reaction to IARC’s decision in 2015 is gathering pace, with moves to discontinue weed control using glyphosate in many jurisdictions, such as local councils and schoolyards. This only amplifies the increased risk from more toxic substitutes, or allergic reactions from uncontrolled weeds producing pollens near schools. Sri Lanka initially banned glyphosate because it feared kidney disease, but on review this decision was reversed for lack of causal evidence and the needs of farmers. Thailand and Vietnam have banned glyphosate more recently because of adverse court decisions in the US, but Thailand also reversed this decision when farmers protested.
Chemical principles are key
It will be chilling for agricultural chemistry and effective pest control if this attack in media and court leads to glyphosate being abandoned without good alternatives. More research has been published on glyphosate generally validating its human and environmental safety than for all other pesticides combined. Even Rachel Carson’s Silent Spring suggested more nuanced technical means of pest control, like making use of the Bacillus thuringiensis peptide crystals toxic to chewing insects. This more subtle approach is now obviating the need for many thousands of tonnes of highly toxic insecticides, by expressing the Bt-toxin genes in GM crops of maize, cotton, potatoes and soybeans. Rachel Carson might even have approved.
The a priori case for suspecting ill of glyphosate, considering all its chemical substituents occur very frequently in biochemical pathways, is weak. No freely soluble zwitterion has been shown as carcinogenic. One can seriously ask whether such a rapidly excreted compound, with all its substituents remaining functional and ionisable in vivo, could be genotoxic. Chemical principles must decide whether this compound, lacking a single feature suggesting mutagenicity, can be seriously carcinogenic. Ultimately, the effects of specific binding activities (or lack of them) with other cell substituents must decide this question. Given the high concentrations used to test tumour formation and even higher levels for genotoxicity, it could be salutary to include common salts as relative controls.
Eventually, given the significant risks to world food supplies and human safety, a rapprochement between the IARC’s methodologies of toxicologists and the regulators of use of agricultural chemicals seems essential.