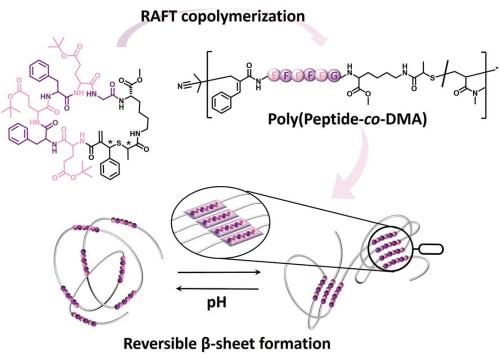
The function of a protein is enabled by its precise 3D structure, which is the result of folding of a sequence-defined polypeptide chain. This hierarchical process starts with the formation of secondary structures (e.g. α-helices and β-sheets) through hydrogen bonding between the backbone amides. The ensemble of secondary structures in turn shapes the overall tertiary structure.
A plethora of strategies has been developed to mimic and control the folding process of synthetic polymers. However, nature’s architectural sophistication remains unmatched, due to the challenging synthesis of long, sequence-defined polymers with functional backbones. Circumventing the need for entirely sequence-defined polymers, work from QUT’s Soft Matter Materials Laboratory led by Hendrik Frisch and PhD student Federica Sbordone reports the use of short peptide sequences that are embedded into the main chain of vinylic polymers to determine the function of the synthetic polymer (Sbordone F., Micallef A., Frisch H. Angew. Chem. Int. Ed. 2024, doi.org/10.1002/anie.202319839). In water, changes in pH lead to the deprotonation/protonation of weakly acidic amino acids, thereby either inducing or disrupting intramacromolecular β-sheet-encoded hydrogen bonding. The pH at which these distinct polymer conformations appear is programmed into the polymer chain by the choice of amino acid sequence and can be synthetically adjusted.