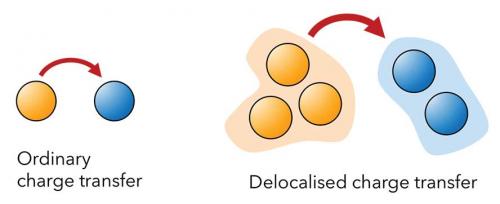
Charge transfer is the simplest chemical reaction, underpinning solar cells, batteries, photosynthesis, combustion, corrosion and molecular electronics. Marcus theory is widely used to describe charge transfer, but is limited to transfer from one donor molecule to one acceptor. However, within many molecular systems – from photosynthetic complexes to organic photovoltaics – charges are coherently delocalised across multiple molecules, making ordinary Marcus theory inapplicable. The only alternatives are simulations that treat the donor and acceptor aggregates as supermolecules, but these are expensive and make it difficult to understand how components of the aggregate contribute to the overall transfer. Researchers at the University of Sydney and the University of Queensland (Taylor N.B., Kassal I. Chem. Sci. 2018, 9, 2942−51) have shown that it is possible to describe charge transfer between delocalised aggregates in terms of the properties of the constituent molecules and couplings between them. The new theory provides qualitative insight into the impact of delocalisation on charge dynamics, predicting effects such as charge supertransfer, a cooperative enhancement of the charge transfer rate above what would be possible with single molecules. This work is expected to clarify charge motion in various molecular systems, including photosynthetic reaction centres, organic photovoltaics and conducting metal–organic frameworks.


The Royal Australian Chemical Institute's magazine for and about the chemical science professions