The overwhelming majority (97%) of atmospheric and climate scientists agree that climate change is happening (doi.org/10.1088/1748-9326/11/4/048002). Yet for more than a decade in Australia, a small but influential minority in science, politics, industry, business and the media continue to promote climate-change scepticism, or even denial, so that Australia has fallen well behind other developed economies in addressing climate change and energy policy.
Despite the scientific consensus, as atmospheric scientists we are often asked by our friends and scientific colleagues ‘Is it real?’ The purpose of this article is to set out the basic observations and physical understanding that show that, yes, it is real, and to provide further resources for readers to access factual, peer-reviewed information.
Simple physics of the greenhouse effect
Try this thought experiment: imagine Earth with its moon in orbit 148 million kilometres from the sun, all three at absolute zero temperature. Now ‘switch on’ the sun with its surface temperature of 5800 K, radiating energy according to Planck’s law with maximum emission intensity in the visible spectrum and providing a total solar energy flux of approximately 1360 W/m2 at the distance of Earth’s orbit. All of this energy reaches the surface of the moon but on Earth approximately one-quarter is scattered back to space by air particles and clouds. The surfaces of Earth and the moon absorb more than 90% of this energy (the rest is reflected), causing them to heat up. The heated surfaces in turn emit radiation back to space (again, Planck’s law, but now with maximum emission in the infrared) until they reach thermal equilibrium where the outgoing infrared radiation balances incoming solar radiation. The moon’s mean surface temperature comes to equilibrium at –18°C, but on Earth the mean surface temperature is +15°C. The difference, 33°C, is principally due to the greenhouse effect. How does this work?
With surface temperatures around 250–300 K, Earth’s and the moon’s emissions are centred in the infrared, maximising at a wavelength near 10 µm. The moon does not have an atmosphere and this infrared radiation escapes unimpeded to space. However, Earth’s atmosphere contains several trace gases that absorb infrared radiation near 10 µm, most important among them being water vapour (H2O), carbon dioxide (CO2), methane (CH4), ozone (O3), nitrous oxide (N2O), and several long-lived halogenated trace gases such as CFC-11 (CCl3F) and sulfur hexafluoride (SF6), collectively referred to as synthetic greenhouse gases. Aerosols also play a role by either absorbing incoming solar radiation or scattering it back to space. Greenhouse gases (GHGs) are transparent to the incoming visible solar radiation but absorb some of the outgoing infrared, thus heating the atmosphere. The net effect is to act as a ‘blanket’, re-emitting some of the absorbed outgoing infrared energy back to the surface and raising the surface temperature. The net greenhouse effect currently warms Earth’s surface relative to the no-atmosphere case (e.g. the moon) by 33°C.
Measuring and modelling the greenhouse effect
Increasing concentrations of GHGs in the atmosphere increase its infrared opacity and further heat Earth’s surface by enhancing the greenhouse effect. The science of GHGs and global warming involves three main components:
- Measuring and understanding the concentrations of the GHGs in the atmosphere, and how they are changing. This is a complex interplay of natural and anthropogenic sources and sinks with atmospheric and oceanic chemistry and biology.
- Calculating the radiative balance between the atmosphere and the surface using global climate models to quantitatively determine both the current temperatures and how temperatures will change under different scenarios of increasing GHG concentrations.
- Understanding the variability of incoming solar radiation both from the sun and due to scattering and absorption of the incoming solar radiation by Earth’s atmosphere.
There is no question that increasing GHG concentrations will lead to higher surface temperatures. The greenhouse effect is real – the scientific challenge is to make accurate calculations of its magnitude and changes into the future. The social and political challenges are much more difficult.
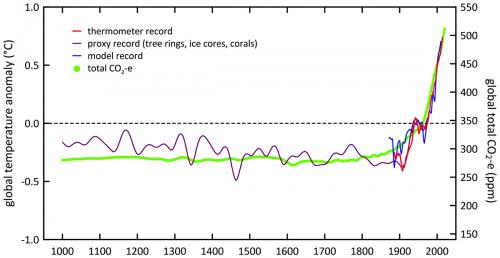
Earth’s surface temperature and GHG levels are increasing together
In response to increasing radiative forcing due to increasing amounts of GHGs, Earth’s surface temperature is increasing. The graph above illustrates the total atmospheric amount of GHGs (as CO2 equivalent (CO2-e)) and Earth’s surface temperature observed and calculated from models incorporating the GHG increase, over the past 1000 years. While the clearly strong correlation does not of itself imply causality, global climate model calculations show that radiative impacts of the increasing GHG amounts are entirely consistent with the observed temperature increase. With current knowledge, no other mechanism can be or needs to be invoked to explain the temperature change.
GHGs and climate forcing
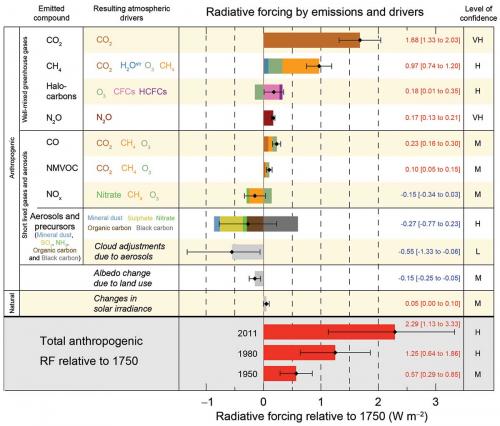
After water vapour, CO2 is the most important GHG, contributing 57% of the climate forcing due to GHGs since pre-industrial times, followed by CH4 (16%), the group of synthetic GHGs (12%) and N2O (5%). These gases are described as long-lived GHGs with atmospheric lifetimes of two years or more, and consequently are relatively well mixed globally.
Tropospheric O3 has an atmospheric lifetime of approximately three weeks and makes up about 10% of the total atmospheric O3 column (~90% is in the stratosphere, 15–40 kilometres above the surface). Tropospheric O3 is described as a short-lived or near-term GHG and its contribution to climate forcing is slightly less than that of CH4.
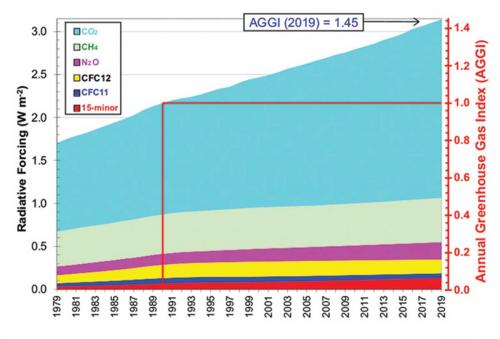
NOAA’s Global Monitoring Laboratory in the US (www.esrl.noaa.gov/gmd) maintains the Annual Greenhouse Gas Index (AGGI), shown in the graph below. The AGGI illustrates both the continuing growth and relative importance of each long-lived GHG to the total. From this graph, we see that total additional radiative forcing in the early 2020s due to all GHGs will be 50% higher than in the reference year of 1990.
Australian contributions to GHG measurements and emissions
CSIRO commenced the first Australian background atmospheric CO2 measurements in the middle-troposphere (5–10 kilometres) over south-east Australia in 1972 and at the surface at Kennaook/Cape Grim, north-west Tasmania (see box) in 1976. These measurements were accompanied at Kennaook/Cape Grim by O3 in 1976, N2O and CFCs in the late 1970s, CH4 in the early-1980s and other synthetic CFCs from 1998; they have continued to the present day.
Observations of Southern Hemispheric GHGs have been extended back 2000 years by measurements on air trapped in Antarctic ice and firn.
In 2018, Australia reported to the United Nations Framework Convention on Climate Change GHG emissions of 537 million tonnes in CO2-e terms (after adjustment for negative emissions due to land use/land use change): 70% CO2, 23% CH4, 4–5% N2O and 2–3% synthetic GHGs.
Carbon dioxide
CO2 has very large two-way exchange fluxes between the atmosphere and Earth’s surface dominated by biosphere uptake (photosynthesis leading to carbon storage in plants and soils) and emissions (respiration by plants and animals). Anthropogenic exchanges of CO2 are tens of times smaller and are dominated by one-way emissions to the atmosphere from fossil fuel use and cement manufacture. Around half of these emissions are taken up by the biosphere and dissolve in the oceans, the remainder stay in the atmosphere. There are also secondary anthropogenic effects through impacts on the biospheric fluxes (e.g. land use change and deforestation). The Global Carbon Project (www.globalcarbonproject.org) provides detailed global accounting for CO2, CH4 and N2O.
CO2 comprises currently (2018) 70% of Australia’s anthropogenic GHG emissions in CO2-e terms, with 95% from the energy sector (stationary and mobile) and 5% from industrial processes. This is the simple consequence of fossil fuel combustion.
Methane
CH4 is the second-most important long-lived GHG after CO2 and is a major source of water vapour and HOx radicals in the stratosphere. CH4 is emitted to the atmosphere roughly equally from natural and anthropogenic sources. The natural sources are predominantly wetlands (30%), the oceans (10%) and lakes and rivers (5%). Wildfires contribute less than 1%. The anthropogenic sources are fossil fuel mining and combustion (15%), ruminants (cattle, sheep etc., 15%), landfills and waste (10%), rice agriculture (5%) and biomass burning (largely biofuels, 5%).
Currently (2018), CH4 makes up 23% of Australia’s GHG emissions in CO2-e terms, with 53% from the agriculture sector, 36% from energy and 11% from waste.
Nitrous oxide
N2O is the third-most important long-lived GHG after CO2 and CH4 and is the dominant source of reactive nitrogen in the stratosphere, contributing significantly to stratospheric O3 chemistry. N2O is emitted to the atmosphere from natural (60%) and anthropogenic (40%) sources. The natural sources are predominantly microbial activity in soils (35%) and the oceans (20%), while the major anthropogenic sources are nitrogenous fertilisers (25%), fossil fuel combustion (5%), biomass burning (5%) and microbial activity in rivers affected by agricultural run-off (3%).
Currently (2018), N2O makes up 4–5% of Australia’s GHG emissions in CO2-e terms, with 72% from agriculture, 16% from the energy sector, 9% from industry and 3% from waste.
Synthetic greenhouse gases
The synthetic GHGs currently (2018) make up 2–3% of Australia’s GHG emissions in CO2-e terms, 100% from the industrial processes sector, 99% from the refrigeration/air conditioning industries (hydrofluorocarbons – HFCs), 1% from aluminium production (perfluorocarbons – PFCs). Many other halogen-containing stratospheric ozone-depleting substances (such as the CFCs) are also effective GHGs.
Tropospheric ozone
Unlike the long-lived GHGs, tropospheric O3 is not emitted into the atmosphere but is formed there primarily through the atmospheric photochemistry of nitrogen oxides and reduced carbon compounds, both of which are emitted into the atmosphere from natural sources and human activities. The main absorption band of O3 is at 9.6 µm near the peak of outgoing radiation and its contribution to climate forcing is slightly less than that of CH4. Tropospheric O3 is the means by which part or all of the radiative forcing of CH4, CO, volatile organic compounds and NOx are realised. Since the start of background measurements in 1896 tropospheric O3 has risen by 30–70% in the mid and high latitudes of the Northern Hemisphere.
Atmospheric aerosols
Atmospheric aerosols contribute to Earth’s radiative balance in two main ways. In the stratosphere, aerosols are formed largely by condensation of water vapour on H2SO4 nuclei, with episodic but significant additions from large volcanic eruptions such as Mt Pinatubo in 1991. Aerosols scatter incoming solar radiation back to space and act to cool Earth’s surface. In the troposphere, aerosols such as black carbon from biomass fires absorb radiation and contribute a positive warming effect. Overall, aerosol contributions to radiative forcing appear to be climate neutral or have a small cooling effect, but with considerable uncertainty.
Changes in ozone-depleting substances, tropospheric ozone and atmospheric aerosols are not accounted for in national GHG inventories but are very important components of global climate change models.
Water vapour
Water vapour is the single most effective GHG, absorbing virtually all radiation between 5.6 and 7.7 µm (1800–1300 cm–1). Atmospheric levels of water vapour are controlled by evaporation, condensation and meteorology rather than any chemical process, and are dependent on feedback from atmospheric and ocean temperatures. Water vapour is not usually included in discussions of increasing GHG amounts due to human activities but the feedback loops between atmospheric and surface temperatures and atmospheric water vapour content are included in all climate change models, approximately doubling the direct temperature increase due to the other GHGs.
The graph on page 22 is from the most recent Intergovernmental Panel on Climate Change report, and presents the relative radiative forcing of the directly emitted GHGs and aerosols.
Atmospheric impacts of the COVID pandemic
The 2020 COVID pandemic significantly reduced atmospheric emissions of GHGs through reduced industrial activity and particularly of CO2 from reduced fossil fuel use in the transport sector. However, it is too early to see a clear impact on atmospheric amounts of CO2 – the reduced 2020 emissions reduce CO2 atmospheric amounts by much less than 1 part per million; this COVID signal is swamped by the natural variability of the many times larger biospheric fluxes.
Simple physics tells us that the greenhouse effect is real, and that increasing levels of GHGs will increase global temperatures. Current global climate models suggest temperature rises of 2–5°C this century, dependent on the scenario chosen for reducing GHG emissions. The human, social and environmental consequences will be immense; adaptation to these changes is another story. Three decades ago, humankind created then avoided a similarly catastrophic problem of global dimension – the threat of increased UV radiation exposure due to stratospheric O3 loss. The problem was avoided with political and industrial will by massively reducing global emissions of O3-depleting halogenated compounds to the atmosphere. Dealing with GHG-induced climate change is a far larger economic, social and technical problem, but addressing the major source, fossil fuel emissions in energy production and transport, is paramount.