This story begins with a chance meeting with three Japanese tourists in Geraldton, in my home state of Western Australia. We met on a family holiday to the north-west of WA, and they had something quite specific on their itinerary. These three young women were determined to see a natural marvel that obviously we didn’t appreciate, having whizzed straight past the turn-off on our way back to Perth. They unfolded a map, pointed to the location of the nearby pink lake and looked at us questioningly. Despite the last stretch to their destination being a long bush-lined road off the main highway and prone to prancing kangaroos between dusk and dawn, they were undeterred. You could see in their eyes that they would do whatever it took to see the pink lake.
The pink lake that these tourists were determined to visit is 100 kilometres north of Geraldton and officially known as Hutt Lagoon. The town of Port Gregory is nestled on its shores, hosting coachloads of international tourists who view the lake by walking, four-wheel-drive and aerial tours. As well as being a tourist drawcard, the pink lake is the site of one of the world’s largest producers of ß-carotene.
Hutt Lagoon – halophile haven
Hutt Lagoon is 12 kilometres by two kilometres and shallow (less than one metre), and classed as a hypersaline evaporative birrida. Birridas are bodies of water that have been isolated over geological time as sea levels decreased. It was an estuary of the nearby Hutt River that became isolated from the sea between coastal dunes during this process. The lagoon receives water from natural rainfall and, because it is below sea level, it also receives marine water by subsurface flow. The salinity range of Hutt River is 3–7 g/L, whereas that of Hutt Lagoon is up to 320 g/L (the range for seawater is 33–37 g/L).
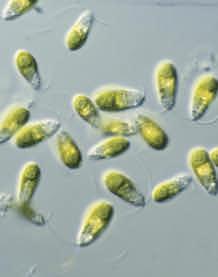
Despite their harshness to ‘normal’ life, saline lakes such as Hutt Lagoon host a variety of salt-tolerant (halophilic) organisms, including crustacea, bacteria and microalgae. Both bacteria and algae can generate pink-red colours in the water, but a delicate balance of water salinity and quality, sunlight and water hydrology optimises the growth of the main actor that causes the pink colour of Hutt Lagoon, the alga Dunaliella salina.
A microalga’s metabolic trick
Pink, orange and red lakes and even red snow have been the object of wonder for centuries, with biblical references in which red-coloured waters are compared to blood: ‘To the Moabites across the way, the water looked red – like blood’ (2 Kings 3:22). It was not until the development of microscopes in the late 16th century that microscopic life was observed and the mystery behind the colour began to be understood.
Prompted by an invitation from the Académie des Sciences in Paris, French botanist Michel Félix Dunal reported what he called Haematococcus salinus and Protococcus in the salterns of Montpellier, France. This study, published in 1838 in Annales des Sciences Naturelles; Botanique, is the first known to identify the alga that now carries Dunal’s name.
Algae are often described in negative terms – think garden pond slimes and toxic algal blooms. But they are vital to marine food webs as well as consuming carbon dioxide and producing oxygen. Algae are simple, non-flowering and typically aquatic plants of a large group that includes the seaweeds and many single-celled forms. Algae contain chlorophyll but lack true stems, roots, leaves and vascular tissue.
D. salina, of the phylum Chlorophyta, is a unicellular, photosynthetic microalga with a distinguishing feature of not having a rigid cell wall. Its chloroplasts, where photosynthesis takes place, are cup shaped. Its cells (each of which is 5–25 micrometres) are ovoid, and each is capable of motion due to two long thin flagella. The cells can ‘swim’ using the flagella with a ‘breaststroke’-type action. D. salina is usually green but develops its red colour with increasing salinity and sunlight, which is often present in saline lakes in arid environments. The alga develops a pink colour to absorb the high-intensity light present as a photoprotective response to chloroplast damage. The compounds that give D. salina its pink-red colour under stressful conditions are a complex mixture of carotenes. The main carotene synthesised is ß-carotene, which the alga achieves, amazingly, by decomposing its green chlorophyll. Levels of 14% dry weight of ß-carotene in D. salina have been reported.
D. salina can survive in saline environments because of another metabolic trick: it synthesises glycerol, maintaining an intracellular glycerol concentration so that the osmotic pressure is higher than the extracellular pressure. This permits the alga to take up water even when growing at high salt concentrations.
Pink lakes change colour naturally with the seasons (rainfall and sunlight), but changes can also be a sign of D. salina’s sensitivity to urbanisation, salt mining and pollutants such as pesticides and heavy metals. An example is the ‘former pink lake’ near the southern WA town of Esperance, in which only the faintest hint remains of its former intense pink colour. Its demise is the object of much study, and a feasibility study of the lake prepared for the Shire of Esperance in 2020 concluded that removal of too much salt has reduced its salinity to a level where D. salina cannot thrive (see executive summary in bit.ly/3tTv0JI). The levels of the alga in this lake have been compared to those in Hutt Lagoon, with the lagoon having 45 times more cells of D. salina per millilitre (p. 20 of feasibility report). The nearby bubble-gum pink Lake Hillier, which is on an island and only accessible for tourist viewing by plane, with aerial tours being available for tourists, is now even more prized and protected by Esperance locals. Although algae are present, recent research indicates the colour is mainly due to the bacterium Salinabacter ruber, which is also able to produce carotenes as photoprotective compounds. Such algal and bacterial halophiles are studied by extremophile scientists to try to predict, among other things, forms of extraterrestrial life.
Carotenoids – more than just a pretty colour
Carotenoids are present in fruits and vegetables, algae and bacteria, and their isoprenoid subunits form a 40-carbon tetraterpenoid backbone. The series of conjugated double bonds is responsible for the molecule’s strong colour: it absorbs light in the blue part of the spectrum (~0.4–0.5 micrometres). The yellow-orange vegetables and fruits are generally α- and ß-carotene rich, while lycopene is responsible for the bright red of tomatoes. Carotenes have many biological functions in humans, who cannot synthesise them. Most importantly, carotenoids are classed as a provitamin A, being converted to vitamin A in the body. Vitamin A is important for normal vision, the immune system, reproduction, and growth and development. The global market for all carotenoids is in the order of US$2.0 billion, promoted by the growing consumer demand for naturally grown products in foods and dietary supplements.
Producing ß-carotene
Hutt Lagoon hosts BASF’s large ß-carotene production facility, comprising 450 hectares of lakes surrounded by 3500 hectares of natural lagoon. BASF has similar facilities in Whyalla in South Australia, making it the world’s largest producer of algal ß-carotene, with its two production sites being the biggest in the world.
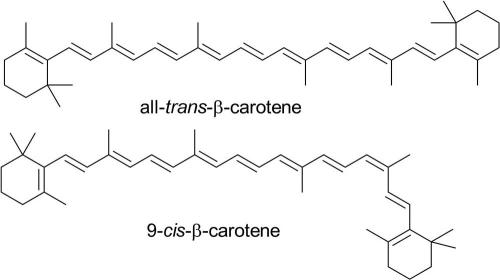
The ß-carotene produced by D. salina is composed of a mixture of the cis- and trans-isomers, with a typical composition of 9-cis (41%), all-trans (42%), 15-cis (10%) and other isomers (6%). ß-Carotene can be produced synthetically, in the all-trans form, but it is 9-cis-ß-carotene that has been used to treat conditions such as retinal dystrophy, chronic psoriasis and atherosclerosis. The cis-isomer is more than 8000 times more expensive, in the pure form, than the trans-isomer.
Much research has been done to optimise the growth of D. salina and the production of ß-carotene. One group of researchers proposed a two-stage process, in which nutrient-rich, lower-salinity nursery ponds were used to boost biomass, with algae then transferred to larger production ponds of higher salinity and with fewer nutrients to promote ß-carotene production (Borowitzka L.J., Borowitzka M.A. Bulletin of Marine Science, 1990, vol. 47(1), p. 244). However, field trials showed this strategy was not feasible: predation by protozoa in the nursery ponds resulted in significant losses. Also, these ponds promoted the preferential growth of other Dunaliella species, and the two-stage process was labour intensive and required a greater pond area. The researchers proposed a semi-continuous process operating at intermediate salinity with optimised nutrient concentrations. The major nutrients added are nitrogen, phosphate and chelated iron. The ratios of calcium to magnesium and chloride to sulfate also affect growth and carotene production.
Harvesting choices and challenges
Harvesting the microscopic algae from large volumes of water poses more technological challenges. Options include filtration using diatomaceous earth, floating fibres on rafts to trap the algae, as well as the use of salinity gradients. The harvested cells may be spray dried to produce an algal powder or extracted with further processing, producing ß-carotene. Various extraction methods, using toluene, hexane or hot oil, can be used. Typical products from the process are spray-dried D. salina for use in animal feeds, including aquaculture, and solutions/suspensions of ß-carotene in vegetable oil for dietary supplements and food colouring.
Supercritical carbon dioxide has also been investigated as an extraction solvent, and optimising pressure and temperature and using a co-solvent have produced good results. The higher energy requirements are offset by the lower solvent costs, with the green properties of carbon dioxide being an extra impetus for the work.
The specific techniques used by different producers are commercial-in-confidence and may be different from those described here (based on research results and other published information). They do, however, give insights into the challenges faced by producers.
D. salina can also be harvested for glycerol – which is used as a solvent, an emollient and a sweetening agent – and for a variety of other carotenoids, lipid components for cosmetic applications as well as proteins and carbohydrates. The use of desalination plant brines to cultivate D. salina, along with the use of carbon dioxide-rich flue gases to promote growth, has also been assessed.
Algal oils and lipids can be used as feedstock for diesel and aviation fuels, while the biomass itself can be converted into biogas and biofuels. An extensive study of WA locations suitable for large-scale microalgal production, such as those with suitable physical locations, light intensities and temperature range, has been completed. The use of microalgae as a renewable energy source continues to be an active area of research and development.