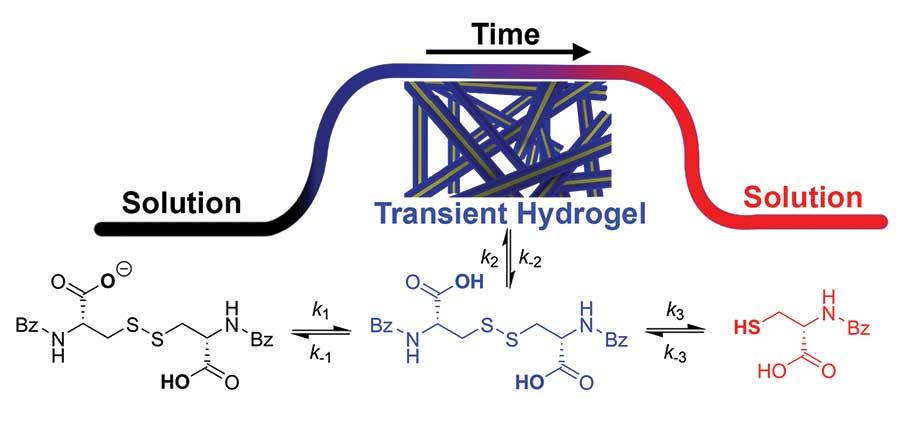
Tubulin and many other self-assembled structures in nature are transient non-equilibrium structures that use some of the free energy provided to form them to perform mechanical work. The synthesis of such a bioinspired transient system based on small molecules has recently been achieved with the development of a transient supramolecular gel by the Thordarson group at the University of New South Wales (Wojciechowski J.P., Martin A.D., Thordarson P. J. Am. Chem. Soc. 2018, 140, 2869−74). The new transient hydrogel system uses orthogonal methods for activation and deactivation, namely the competition between the gelation of the oxidised cystine form of the N,N’-dibenzyol-L-cystine (DBC) gelator and its reduction to the soluble cysteine by a chemical reducing agent, tri(2-carboxyethyl)phosphine (TCEP). Detailed kinetic analysis of the system allowed the researchers to program the lifetime of the gels with good accuracy down to 15 minutes. Furthermore, the team could generate multiple transient hydrogel cycles (sol↔gel oscillations) simply by periodically adding more of the DBC gelator to a TCEP ‘bath’. The general concept of this work is applicable not only to redox-active systems but to systems responsive to other stimuli, which should aid the design of next-generation spatiotemporal hydrogel materials.


The Royal Australian Chemical Institute's magazine for and about the chemical science professions