Polarising sunglasses are very common, but do we really know what makes them tick? We know they reduce glare because some annoying reflected light is polarised and can be blocked, but results in funny colours when looking at car windscreens and LCD displays. A sheet of polaroid transmits only light waves that align with the filter in the sunglasses.
When light enters materials
On a summer’s day by the pool, you’ve probably noticed (if you take your sunglasses off) that the legs of people in the pool appear to bend upwards. This is explained by a physical property called refractive index – the light beam changes direction when entering the water from the air. The amount of change differs between materials and decreases with the wavelength (blue to red) of light. That’s how a glass prism works.
A material can have different refractive indices in different orientations, such as some plastic materials that have been stretched in one direction. For other materials, such as crystals, this is a natural property. The difference between these refractive indices is called the birefringence of the material.
What you need
- 2 pieces of polaroid (such as the plastic in polaroid sunglasses)
- Glass microscope slide
- Clear sticky (adhesive) tape
- Sunlight or other light source
What to do
- Cover the centre of the microscope slide lengthways with a section of clear sticky tape.
- Place a slightly shorter length of sticky tape over the first piece, leaving a small gap at one end. Continue making this ‘step ladder’ for around ten steps.
- Position your pieces of polaroid at 90° to each other.
- Place your ‘step ladder’ between the crossed polarisers. A series of different colours should appear with each step. If not, make each step twice as thick or try another type of tape.
What’s happening?
Tape is a birefringent material, and gradually increasing its thickness produces a sequence of colours as different wavelengths in turn are transmitted by the crossed (second, called analysing) polaroid. The colour sequence for crossed polarisers is, roughly, yellow, blue, magenta, lime green, purple, pale green, colourless.
When you have this working, rotate one of the polarisers gradually to 45° so that their complementary colour partners take over. You may even be able to get a second-order series higher up the ladder.
For a more detailed explanation of this complicated phenomenon see https://physlab.lums.edu.pk/imagesc/ce/Cellophane1.pdf. For an explanation of the relationship between the speed of light and refractive index with that wonderful ‘principle of least action’, see a familiar reference (*B. Selinger, R. Barrow, Chemistry in the marketplace CSIRO Publishing, 2017, 6th edn, pp. 483–4).
Molecules rotating light
Certain molecules in solution can rotate the plane of (circularly) polarised light that travels in a corkscrew fashion, moving forward and rotating at the same time, either clockwise or anticlockwise.
What you need
- Sucrose (plain white sugar), glucose and fructose (available from health stores or bulk pharmacies)
- 2 pieces of polaroid
- Torch with LED globe
- Test tubes or tall (narrow) jars
What to do
- Prepare solutions of sucrose, glucose and fructose by dissolving approximately three heaped tablespoons of each in half a (measuring) cup (125 mL) of water.
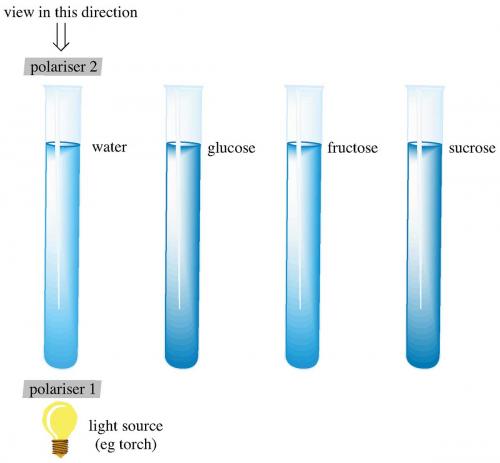
- Three-quarters fill a test tube with each of the solutions and a fourth one with just water.
- Line up the apparatus vertically as shown, with the light source at the bottom, followed a by a polariser, then a test tube, then a second (analyser) polariser.
- With pure water, rotate the analyser polariser to the point of maximum light extinction. Mark the edge of both polaroid sheets so you will know how much they are rotated in the next steps. Replace the water-containing test tube with each sugar solution in turn.
- Light will come through the previously extinguished crossed polarisers. Rotate the analyser polariser to reach (approximate) extinction again.
- One colour at a time (as seen in the sticky tape experiment) comes through. For sucrose and glucose, the rotation needed will be clockwise and for fructose it will be anticlockwise.
- For ease, adjust the sugar concentrations and/or the depth of solution in the test tubes so that the rotation angles needed are around 20°.
What’s happening?
The optical rotation caused by the sugar molecules is due to (circular) birefringence. The refractive indices (speed of light) of left-hand circularly polarised light and right-hand circularly polarised light are different. The ability of these sugar molecules to do this is a property (optical activity) of their molecular geometry.
Chemists use the single-colour sodium D-line (as seen with sodium vapour street lights) to obtain sharp extinctions. By using a (white) LED torch we do the opposite.
Sugar Optical rotation*.
Sucrose +66°
Glucose (dextrose) +53°
Fructose –92°
*Relative rotation measured with yellow sodium D-line at 589 nm.
When sugar (sucrose) is broken into its two components glucose and fructose, the optical rotation reverses from +66° to –39°, and so this (roughly twice as sweet) mixture is known as invert sugar! Honey is a ‘natural’ example.
Acknowledgements
Thanks to Emeritus Professor Elmars Krausz (ANU) for discussions on this and related optical topics and to Jenny Selinger (Head of Science, Emanuel College, Sydney) for comments and testing.