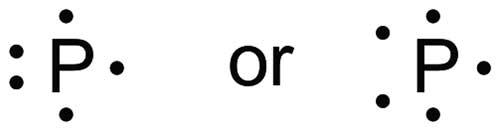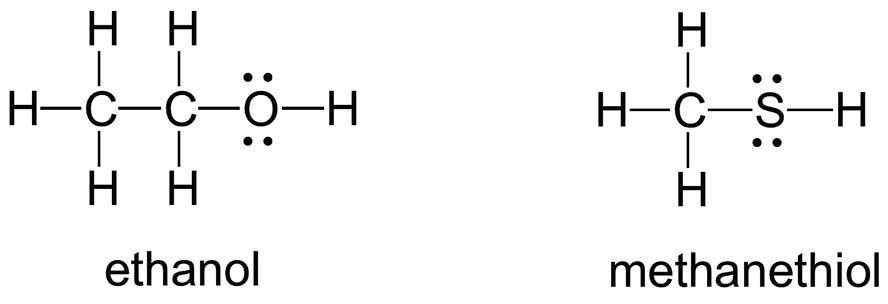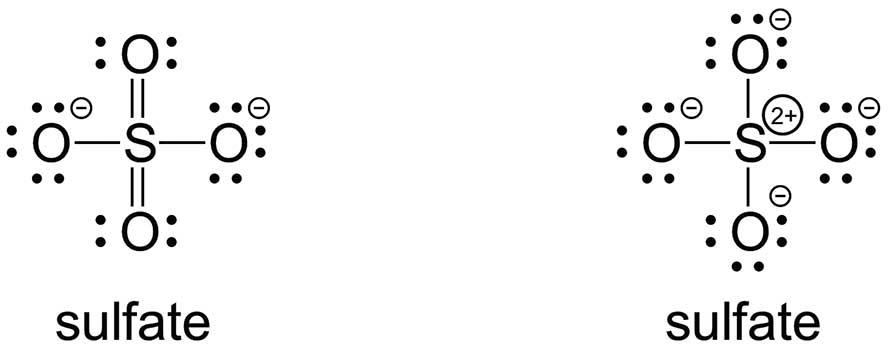Lewis structures are a key concept in chemistry education because they can be used to predict molecular geometries, relative bond lengths and bond dissociation energies, aromaticity and other properties. They can also be used to confirm if the molecular and ionic composition of a chemical formula is reasonable.
Lewis structures are representations that explicitly show all atoms, all valence electrons and formal charges. The valence electrons are usually arranged in pairs, which are either localised on particular atoms or localised between a pair of adjoining atoms. Bonding pairs of electrons are usually denoted by line segments, representing covalent bonds. A double bond is represented by a pair of parallel line segments, while a triple bond is represented by three parallel line segments. Formal charges of atoms indicate the difference between the positive nuclear charge and the total number of (core and valence) electrons, on the formal basis that bonding electrons are shared equally between atoms they join.
One common method is to assemble Lewis structures from the Lewis symbols for individual atoms:
This method is confusing for students when hypervalent atoms are involved:
A second common method is a multistep algorithmic approach that counts valence electrons, arranges atoms and distributes electrons to form bonds and lone pairs, in order to either fulfil the ‘octet rule’ or minimise formal charge.
Lewis structures such as HSO4−, C2H3O2− and CO, with multiple bonds or charges, are problematic for students who are new to Lewis structures. Problems associated with drawing structures with more than one central atom (e.g. N2F5+, P2S3, HSO4− and C2H3O2−) or with an atom from beyond the second row of the periodic table (e.g. S, P, Br and I) often persist into second-year undergraduate studies. In one research study with a group of second-year organic-chemistry students, around 80% of students were able to draw correct Lewis structures with one carbon atom (e.g. CH2O, HCN, CH3OH, CH6N+ and CH5O+), but only around 30% were able to draw correct structures with two or more carbon atoms (e.g. CH3COOH, C2H5O– and C2H3O2–). The ability to draw a correct Lewis structure can depend on how information is presented: more than 90% of the group were able to draw the structure for methanol when given the condensed formula (CH3OH), but only around 60% could when given the molecular formula (CH4O). When asked to draw structures for C2H6O and CH5S, students often prefer symmetrical or ‘balanced’ structures:
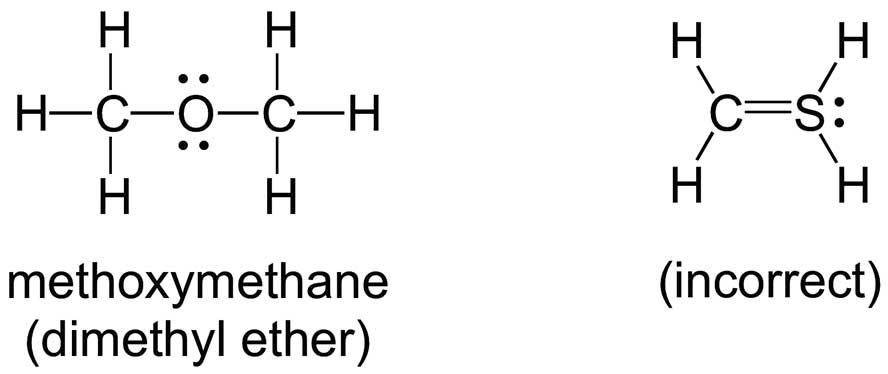
and felt uncomfortable with unsymmetrical structures:
Educators also have problems with Lewis structures, but for different reasons. An ‘octet-rule’ Lewis structure for sulfate anion would have four S–O single bonds, while a ‘minimal-formal-charge’ structure would have an ‘expanded octet’, which is usually explained by invoking the involvement of d-orbitals in the bonding:
Note that only five elements (C, N, O, F and Ne) always obey the octet rule of having eight valence bonding and lone-pair electrons: all the other elements can have ‘depleted octets’ or ‘expanded octets’. Argon compounds have been synthesised and have expanded octets.
Some researchers argue that the expanded-octet structures of the oxides, oxoacids and oxoanions of the 3p–5p non-metals (e.g. P, As, S, Se, Te, Cl, Br, I and Xe) are more consistent with experimental bond lengths than structures that obey the octet rule. Other researchers point out that expanded octets are unrealistic because the energies of the d-orbitals are too high to be involved in bonding.
Some quantum chemistry studies show that the electron densities of a series of P–O, S–O and Cl–O bonds are more consistent with expanded octets, while other studies show that the calculated molecular orbitals are more consistent with the octet rule.
Most recently, quantum chemistry studies have shown the valence s- and p-electrons can form ‘recoupled pair bonds’ instead of lone pairs (see Takeshita et al. J. Phys. Chem. A, 2015, vol. 119, pp. 7683–94). It is therefore possible to form up to four bonds for carbon and six bonds for sulfur. It would seem that the approach of minimising formal charges where possible is correct, but without involvement of the d-orbitals.
Both the single-bonded octet-rule and the hexavalent minimal-formal-charge structures for sulfate would lead students to predict a tetrahedral geometry using VSEPR theory. Ultimately, we should remember that Lewis structures are pictorial representations or models; they are not reality. Like all tools, Lewis structures are useful constructs: we should teach Lewis structures for their predictive utility, and not as an end in itself. And we should place greater emphasis on the drawing of structures with more than one central atom and structures with at least one atom with an expanded octet. We should also ban the term ‘octet rule’.