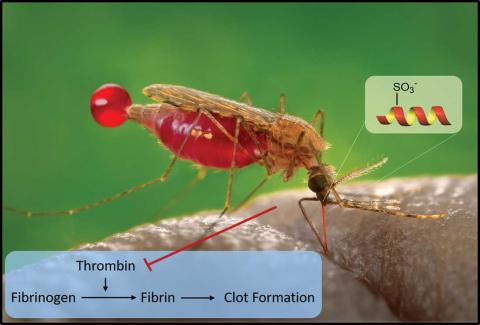
Many blood-feeding organisms, including the malaria mosquito vector Anopheles, possess a suite of anticoagulant proteins in their saliva to enable access to their blood meal. Many of these proteins target the central blood-clotting enzyme thrombin, with this interaction modulated by post-translational modifications of the salivary proteins. Post-translational sulfation of tyrosine residues has been predicted to be important for activity, but such modifications are notoriously difficult to detect owing to the extremely labile nature of the aryl sulfate ester modification. The Payne laboratory at the University of Sydney has led a multidisciplinary effort to determine the functional role of this modification in salivary proteins from the Anopheles mosquito called the anophelins and demonstrates the potential application of this family of proteins as therapeutic anticoagulants (Watson E.E., Liu X., Thompson R.E., Ripoll-Rozada J., Wu M., Alwis I., Gori A., Loh C.-T., Parker B.L., Otting G., Jackson S., Pereira P.J.B., Payne R.J. ACS Cent. Sci. 2018, 4, 468–76). Using cutting-edge peptide ligation methods, the team synthesised a library of differentially sulfated anophelin proteins in homogeneous form. In vitro biochemical screening of these sulfoproteins demonstrated the importance of modification at these key residues, with sulfation yielding up to two orders of magnitude increase in inhibition of thrombin. Most excitingly, these sulfoproteins were shown to prevent clot formation in an in vivo model of thrombosis, suggesting that they may have clinical significance for diseases such as stroke.
CDC/James Gathany/CC-PD-Mark


