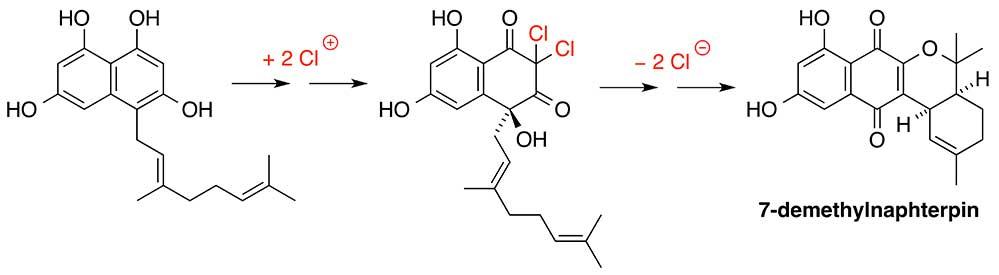
Marine microorganisms have evolved vanadium-dependent chloroperoxidase (VCPO) enzymes that are capable of oxidising chloride anions in the ocean into an electrophilic source of ‘Cl+’. This allows chlorine to be introduced into organic molecules in the biosynthesis of chlorinated natural products. Recent work conducted by researchers at the University of Adelaide and the Scripps Institution of Oceanography, USA, has shown that marine Streptomyces bacteria also use VCPO enzymes to oxidise aromatic ring systems by cryptic chlorination in the biosynthesis of the naphterpin and marinone families of meroterpenoid natural products (Murray L.A.M, McKinnie S.M.K, Pepper H.P., Erni R., Miles Z.D., Cruickshank M.C., Lopez-Perez B., Moore B.S., George J.H. Angew. Chem. Int. Ed. 2018, 57, 11 009–14). The biosynthetic sequence is initiated by VCPO-mediated oxidative dearomatisation and dichlorination, followed by an a-hydroxyketone rearrangement. Subsequent loss of chloride-anion leaving groups allows the formation of a highly oxidised naphthoquinone core. The entire biosynthetic pathway was mimicked in biomimetic total syntheses of 7-demethylnaphterpin and debromomarinone. Several proposed biosynthetic intermediates were also synthesised and used as substrates and synthetic standards to help elucidate the function of two new VCPO enzymes, MarH1 and MarH3, that initiate marinone biosynthesis.


The Royal Australian Chemical Institute's magazine for and about the chemical science professions