The Royal Swedish Academy of Sciences has awarded the Nobel Prize in Chemistry 2022 to Carolyn R. Bertozzi (Stanford University, USA), Morten Meldal (University of Copenhagen, Denmark) and K. Barry Sharpless (Scripps Research, La Jolla, USA) ‘for the development of click chemistry and bioorthogonal chemistry’.
The Nobel Prize in Chemistry 2022 is about making difficult processes easier. Sharpless and Meldal have laid the foundation for a functional form of chemistry – click chemistry – in which molecular building blocks snap together quickly and efficiently. Bertozzi has taken click chemistry to a new dimension and started using it in living organisms.
Chemists have long been driven by the desire to build increasingly complicated molecules. In pharmaceutical research, this has often involved artificially recreating natural molecules with medicinal properties. This has led to many admirable molecular constructions, but these are generally time consuming and very expensive to produce.
‘This year’s Prize in Chemistry deals with not overcomplicating matters, instead working with what is easy and simple. Functional molecules can be built even by taking a straightforward route,’ said Johan Åqvist, Chair of the Nobel Committee for Chemistry.
Sharpless – who is now being awarded his second Nobel Prize in Chemistry – started the ball rolling. Around the year 2000, he coined the concept of click chemistry, which is a form of simple and reliable chemistry, where reactions occur quickly and unwanted by-products are avoided.
Shortly afterwards, Meldal and Sharpless – independently of each other – presented what is now the crown jewel of click chemistry: the copper-catalysed azide–alkyne cycloaddition. This is an elegant and efficient chemical reaction that is now in widespread use. Among many other uses, it is utilised in the development of pharmaceuticals, for mapping DNA and creating materials that are more fit for purpose.
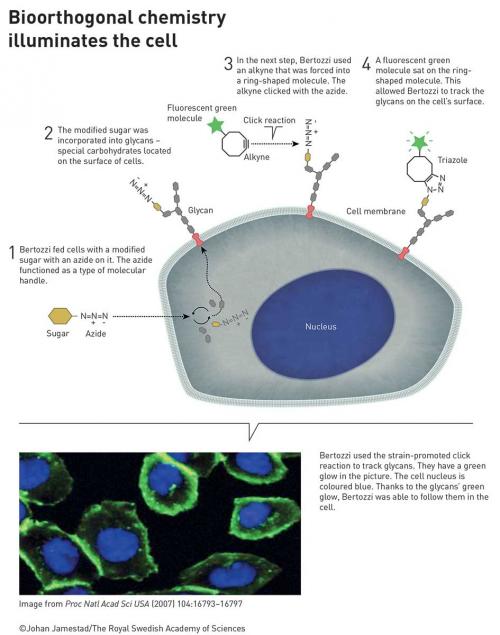
Bertozzi took click chemistry to a new level. To map important but elusive biomolecules on the surface of cells – glycans – she developed click reactions that work inside living organisms. Her bioorthogonal reactions take place without disrupting the normal chemistry of the cell.
These reactions are now used globally to explore cells and track biological processes. Using bioorthogonal reactions, researchers have improved the targeting of cancer pharmaceuticals, which are now being tested in clinical trials.
Click chemistry and bioorthogonal reactions have taken chemistry into the era of functionalism. This is bringing the greatest benefit to humankind.