Physiology or Medicine: immune defence in cancer treatment
The Nobel Prize in Physiology or Medicine 2018 was awarded jointly to James P. Allison and Tasuku Honjo ‘for their discovery of cancer therapy by inhibition of negative immune regulation’.
Cancer kills millions of people every year and is one of humanity’s greatest health challenges. By stimulating the inherent ability of our immune system to attack tumour cells, this year’s Nobel laureates have established an entirely new principle for cancer therapy.
James P. Allison studied a known protein that functions as a brake on the immune system. He realised the potential of releasing the brake and thereby unleashing our immune cells to attack tumours. He then developed this concept into a new approach for treating patients.
In parallel, Tasuku Honjo discovered a protein on immune cells and, after careful exploration of its function, eventually revealed that it also operates as a brake, but with a different mechanism of action. Therapies based on his discovery proved to be strikingly effective in the fight against cancer.
Allison and Honjo showed how different strategies for inhibiting the brakes on the immune system can be used in the treatment of cancer. The seminal discoveries by the two laureates constitute a landmark in our fight against cancer.
Can our immune defence be engaged for cancer treatment?
Cancer comprises many different diseases, all characterised by uncontrolled proliferation of abnormal cells with capacity for spread to healthy organs and tissues. A number of therapeutic approaches are available for cancer treatment, including surgery, radiation and other strategies, some of which have been awarded previous Nobel Prizes. These include methods for hormone treatment for prostate cancer (Huggins 1966), chemotherapy (Elion and Hitchins 1988) and bone marrow transplantation for leukaemia (Thomas 1990). However, advanced cancer remains immensely difficult to treat, and novel therapeutic strategies are desperately needed.
In the late 19th century and beginning of the 20th century, the concept emerged that activation of the immune system might be a strategy for attacking tumour cells. Attempts were made to infect patients with bacteria to activate the defence. These efforts only had modest effects, but a variant of this strategy is used today in the treatment of bladder cancer. It was realised that more knowledge was needed. Many scientists engaged in intense basic research and uncovered fundamental mechanisms regulating immunity and also showed how the immune system can recognise cancer cells. Despite remarkable scientific progress, attempts to develop generalisable new strategies against cancer proved difficult.
Accelerators and brakes in our immune system
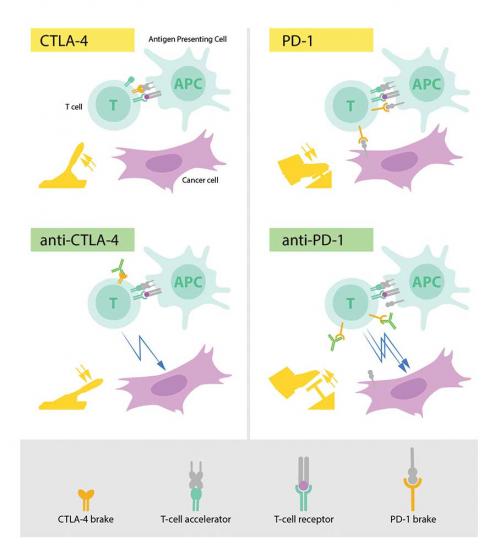
The fundamental property of our immune system is the ability to discriminate ‘self’ from ‘non-self’ so that invading bacteria, viruses and other dangers can be attacked and eliminated. T cells, a type of white blood cell, are key players in this defence. T cells were shown to have receptors that bind to structures recognised as non-self and such interactions trigger the immune system to engage in defence. But additional proteins acting as T-cell accelerators are also required to trigger a full-blown immune response (see diagram p. 7). Many scientists contributed to this important basic research and identified other proteins that function as brakes on the T cells, inhibiting immune activation. This intricate balance between accelerators and brakes is essential for tight control. It ensures that the immune system is sufficiently engaged in attack against foreign microorganisms while avoiding the excessive activation that can lead to autoimmune destruction of healthy cells and tissues.
A new principle for immune therapy
During the 1990s, in his laboratory at the University of California, Berkeley, James P. Allison studied the T-cell protein CTLA-4. He was one of several scientists who had made the observation that CTLA-4 functions as a brake on T cells. Other research teams exploited the mechanism as a target in the treatment of autoimmune disease. Allison, however, had an entirely different idea. He had already developed an antibody that could bind to CTLA-4 and block its function (see diagram p. 7). He now set out to investigate if CTLA-4 blockade could disengage the T-cell brake and unleash the immune system to attack cancer cells. Allison and co-workers performed a first experiment at the end of 1994, and in their excitement it was immediately repeated over the Christmas break. The results were spectacular. Mice with cancer had been cured by treatment with the antibodies that inhibit the brake and unlock antitumour T-cell activity. Despite little interest from the pharmaceutical industry, Allison continued his intense efforts to develop the strategy into a therapy for humans. Promising results soon emerged from several groups, and in 2010 an important clinical study showed striking effects in patients with advanced melanoma, a type of skin cancer. In several patients, signs of remaining cancer disappeared. Such remarkable results had never been seen before in this patient group.
Discovery of PD-1 and its importance for cancer therapy
In 1992, a few years before Allison’s discovery, Honjo discovered PD-1, another protein expressed on the surface of T cells. Determined to unravel its role, he meticulously explored its function in a series of elegant experiments performed over many years in his laboratory at Kyoto University. The results showed that PD-1, similar to CTLA-4, functions as a T-cell brake, but operates by a different mechanism (see diagram p. 7). In animal experiments, PD-1 blockade was also shown to be a promising strategy in the fight against cancer, as demonstrated by Honjo and other groups. This paved the way for utilising PD-1 as a target in the treatment of patients. Clinical development ensued, and in 2012 a key study demonstrated clear efficacy in the treatment of patients with different types of cancer. Results were dramatic, leading to long-term remission and possible cure in several patients with metastatic cancer, a condition that had previously been considered essentially untreatable.
Immune checkpoint therapy for cancer today and in the future
After the initial studies showing the effects of CTLA-4 and PD-1 blockade, the clinical development has been dramatic. We now know that the treatment, often referred to as ‘immune checkpoint therapy’, has fundamentally changed the outcome for certain groups of patients with advanced cancer. Similar to other cancer therapies, adverse side effects are seen, which can be serious and even life threatening. They are caused by an overactive immune response leading to autoimmune reactions, but are usually manageable. Intense continuing research is focused on elucidating mechanisms of action, with the aim of improving therapies and reducing side effects.
Of the two treatment strategies, checkpoint therapy against PD-1 has proved more effective, and positive results are being observed in several types of cancer, including lung cancer, renal cancer, lymphoma and melanoma. New clinical studies indicate that combination therapy, targeting both CTLA-4 and PD-1, can be even more effective, as demonstrated in patients with melanoma. Thus, Allison and Honjo have inspired efforts to combine different strategies to release the brakes on the immune system with the aim of eliminating tumour cells even more efficiently. A large number of checkpoint therapy trials are currently underway against most types of cancer, and new checkpoint proteins are being tested as targets.
For more than 100 years, scientists attempted to engage the immune system in the fight against cancer. Until the seminal discoveries by the two laureates, progress into clinical development was modest. Checkpoint therapy has now revolutionised cancer treatment and has fundamentally changed the way we view how cancer can be managed.
Physics: tools made of light
The Nobel Prize in Physics 2018 was awarded with one half to Arthur Ashkin ‘for groundbreaking inventions in the field of laser physics’, and the other half jointly to Gérard Mourou and Donna Strickland ‘for their method of generating high-intensity, ultra-short optical impulses’.
The inventions being honoured this year have revolutionised laser physics. Extremely small objects and incredibly rapid processes are now being seen in a new light. Advanced precision instruments are opening up unexplored areas of research and a multitude of industrial and medical applications.
Ashkin invented optical tweezers that grab particles, atoms, viruses and other living cells with their laser beam fingers. This new tool allowed Ashkin to realise an old dream of science fiction – using the radiation pressure of light to move physical objects. He succeeded in getting laser light to push small particles towards the centre of the beam and to hold them there. Optical tweezers had been invented.
A major breakthrough came in 1987, when Ashkin used the tweezers to capture living bacteria without harming them. He immediately began studying biological systems and optical tweezers are now widely used to investigate the machinery of life.
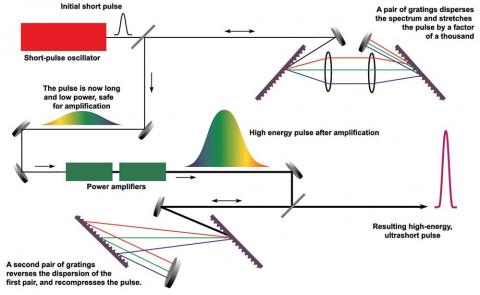
Mourou and Strickland paved the way towards the shortest and most intense laser pulses ever produced. Their revolutionary article was published in 1985 and was the foundation of Strickland’s doctoral thesis.
Using an ingenious approach, they succeeded in creating ultrashort high-intensity laser pulses without destroying the amplifying material. First, they stretched the laser pulses in time to reduce their peak power, then they amplified them, and finally they compressed them. If a pulse is compressed in time and becomes shorter, then more light is packed together in the same tiny space – the intensity of the pulse increases dramatically.
Strickland and Mourou’s newly invented technique, called chirped pulse amplification (CPA), soon became standard for subsequent high-intensity lasers. Its uses include the millions of corrective eye surgeries that are conducted every year using the sharpest of laser beams.
The innumerable areas of application have not yet been completely explored. However, even now these celebrated inventions allow us to rummage around in the microworld in the best spirit of Alfred Nobel – for the greatest benefit to humankind.
US Department of Energy
Chemistry: harnessing the power of evolution
The Nobel Prize in Chemistry 2018 was awarded with one half to Frances J. Arnold ‘for the directed evolution of enzymes’, and the other half jointly to George P. Smith and Gregory P. Winter ‘for the phage display of peptides and antibodies’.
The power of evolution is revealed through the diversity of life. The 2018 Nobel laureates in Chemistry have taken control of evolution and used it for purposes that bring the greatest benefit to humankind. Enzymes produced through directed evolution are used to manufacture everything from biofuels to pharmaceuticals. Antibodies evolved by a method called phage display can combat autoimmune diseases and, in some cases, cure metastatic cancer.
Since the first seeds of life arose around 3.7 billion years ago, almost every crevice on Earth has filled with different organisms. Life has spread to hot springs, deep oceans and dry deserts, all because evolution has solved a number of chemical problems. Life’s chemical tools – proteins – have been optimised, changed and renewed, creating incredible diversity.
This year’s Nobel laureates in Chemistry have been inspired by the power of evolution and used the same principles – genetic change and selection – to develop proteins that solve human’s chemical problems.
In 1993, Frances H. Arnold conducted the first directed evolution of enzymes. Since then, she has refined the methods that are now routinely used to develop new catalysts. The uses of Arnold’s enzymes include more environmentally friendly manufacturing of chemical substances, such as pharmaceuticals, and the production of renewable fuels for a greener transport sector.
In 1985, George Smith developed an elegant method known as phage display, where a bacteriophage – a virus that infects bacteria – can be used to evolve new proteins. Gregory Winter used phage display for the directed evolution of antibodies, with the aim of producing new pharmaceuticals. The first one based on this method, adalimumab, was approved in 2002 and is used for rheumatoid arthritis, psoriasis and inflammatory bowel diseases. Since then, phage display has produced antibodies that can neutralise toxins, counteract autoimmune diseases and cure metastatic cancer.
We are in the early days of directed evolution’s revolution, which, in many different ways, is bringing and will bring the greatest benefit to humankind.
(These laureates and their work will be featured in Chemistry in Australia early in 2019.)