In 1876, there were no effective treatment options for common bacterial infections, which were often lethal in the era before antibiotics. That year, John Tyndall published his findings that penicillin killed bacteria but his paper was ignored. In 1929, Alexander Fleming published his finding on penicillin and likewise it was initially ignored – until 1938 when Howard Florey came across his paper and decided to act. Until Florey proved otherwise, the prevailing attitude was that antibacterial drugs were a delusion. A similar attitude prevails today in relation to cures for neuroplasticity-related issues.
Until recently, neuroplasticity – the brain’s ability to re-wire itself, in effect altering its own genetic operation – was firmly believed to be non-existent and impossible in adults. Like many widely held beliefs, it has now been shown to be false. We now know that many serious conditions could potentially be relieved or remedied by promoting, assisting and/or guiding the brain to re-wire itself and yet it is still a prevalent widely held conviction that drugs directing neuroplastic changes are a delusion.
The harsh reality is that there are no effective pharmacological means at present to neither significantly assist nor reverse existing damage, let alone cure the underlying causes of a wide array of intellectual disability, dementias, neurodevelopmental disorders (including autism and Down syndrome), age-dependent neurodegenerative diseases (including Alzheimer’s and Parkinson’s disease) and many other neurological-related applications.
Current pharmacological tools in psychiatry are effectively limited to symptom relief but are not in themselves curative. The current drugs often help and are needed for management and for other therapies to have a chance to work (e.g. psychotherapy). Schizophrenias, OCD, ADHD, anxieties and phobias, damage due to child abuse, neglect and trauma as well as many others, all potentially fall within the scope as being amenable to neuroplastic pharmacological curative tools yet to be created.
The potential scope of applications is much wider and can apply to healthy normal people such as enhancement of learning abilities (similar to that seen in very young children).
In the wider community (as well as within professional circles) there is a widely held hope that one day a ‘miracle’ discovery will be made and then these issues will be adequately dealt with. Reality and historical precedents say otherwise.
The disconnect between research and application, and thus a hurdle on the path to change, can be considered in terms of the maturity of technology, or technology readiness level (TRL).
The mythology that medical researchers on their own and/or pharmaceutical giants will make this happen is fervently clung to by many.
No one in the private sector paid any attention to Tyndall’s (TRL1) nor Fleming’s (TRL1) work until Howard Florey serendipitously found Fleming’s paper and showed that it works in practice having directed the efforts that isolated the active compound, purified it and manufactured in sufficient quantities to conduct initial clinical experiments (TRL3). Only then did industry (prodded heavily by the US government of the day due to its projected immediate wartime value) come on board and in record time bring it to TRL9 within four years. Nominally it takes 10–20 years to transition drugs from TRL3 to TRL9.
A golden opportunity for chemists in academia
Without chemists, especially tenured academics who in spirit and position are similar to Florey (who was able and willing to obtain and commit resources and skills to try something absolutely new and unproven) taking a leading role, medical theories and discoveries (TRL0–2) will very likely continue to be made but history continues to repeat as these are frequently seemingly ignored because they are not a viable commercial product (TRL9 or at the very least TRL3) as yet.
History and current practice remains that pharmaceutical companies do follow medical research and do conduct their own research internally (and/or sponsored); however, there is no denying that the industry has historically and consistently shown an apparently very strong preference to ‘ambulance chase’. To minimise their risk envelope, they wait for someone else to make a commercially valuable proven product and then buy them out, copy them or use that as a ‘lead’ to either explore or use combinatorial libraries to try structurally related analogues. The prominent firms at the time all missed the boat in terms of both opportunity and staggering profits with cytokines (the genetic signalling molecules initially comprising G-CSF and GM-CSF) for cancer applications because it was outside the usual expectations for pharmacological entities.
Attitudes matter
The limiting step in paradigm change is perceptions and attitudes. If you fervently believe that it can’t be done, then why would you even try?
It would be very reasonable to assert that unless another ‘Florey’ comes along and takes the results of existing medical research results and transitions one or more of these into working prototype(s) that will prove the concept (at least to TRL3), then it is highly unlikely we will see the oft-hoped-for ‘miracle cure(s)’.
To date, very little attention is seemingly being paid by applied chemists to a mountain of high-quality data (mostly TRL1) that hints at appropriate targets for drug design and development in the areas of dementias, neurodevelopmental disorders, psychological and neurological disorders, blindness, deafness and learning issues, which are all associated with neuroplasticity. Few chemists in academia and industry take much notice of research outside of their chosen areas of interest, and genetic research is often viewed as not being ‘chemically relevant’.
Genetic experiments reveal targets for drug design
Oncology (cancer) research has made major contributions to date, but perhaps the most overlooked contribution is also the most important.
Oncogenes are an inherently inbuilt system of ‘master regulator genes’. When these go faulty, the result is cancer. They exist to control, monitor and regulate the operation of all other genes. They do this using a system of receptors and messenger/signalling molecules. Thus, any research that shows activity/benefit by switching ‘on’ or ‘off’ various genes by means of genetic experiments using vectors (genetically modified viruses that either introduce and paste code or cut code out or a mix thereof) can translate to targets for drug design and development.
Every successful genetic experiment medical researchers report is an alert to the existence of targets that are pharmacologically amenable to getting the same end result using appropriately designed drugs. The precise biochemical mechanisms may not be known as yet, but there is potential to design drugs that either mimic or duplicate the messages or selectively block the relevant receptors. The key to success is knowing what the targets actually are.
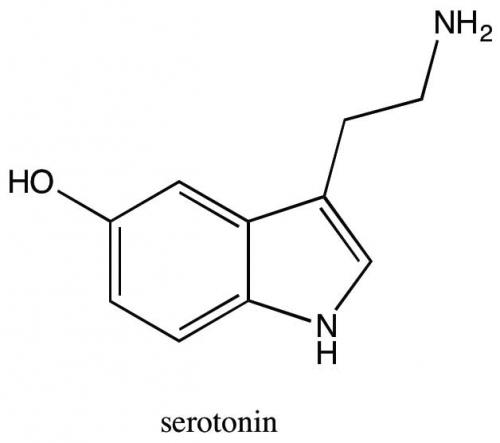
Knowing the precise and correct target is critical: the case of serotonin
Serotonin (5-hydroxytryptamine or 5-HT) modulates cortical function via widely projecting axons, which act on a diversity of receptor subtypes. Serotonin receptors are highly heterogeneous and they have been regrouped within seven different families (5-HT1–5-HT7). With the exception of the 5-HT3 family, which is a ligand-gated ion channel, all others are G-protein coupled receptors, with each family sharing structural, pharmacological and transductional characteristics. 5-HT receptors have been implicated in the regulation of several psychiatric and neurological disorders related to serotonergic neurotransmission, and specific receptor subtypes have recently been associated with either the pathogenesis or the treatment of migraine headache. Current evidence continues to support the use of selective serotonin reuptake inhibitors as first-line pharmacologic interventions for OCD. Neuroplasticity modulation and enhancement via 5-HT agonists may well have a very useful role to play in chronic fatigue syndrome, fibromyalgia and irritable bowel syndrome as well as other implicated medically unexplained symptoms.
Following the amazing success of 5-HT3 agonist ondansetron at changing the face of medical treatment of nausea and vomiting, attention turned to a spectrum of 5-HT receptors as potentially useful targets for drug design and development.
Between 2000 and 2007 Glaxo (UK) published their efforts to develop a novel (hopefully blockbuster class) rapid-acting antidepressant, having pinned their hopes and efforts on serotonin reuptake via a well-designed agonist initially for 5-HT1B, then a dual-acting 5-HT1A/5-HT1B agonist (TRL5). A recent paper by Melburnian medical researchers suggest they may well have had far better results if they targeted 5-HT2A receptors instead. It’s now been demonstrated that 5-HT2A agonists enhance functional connectivity between hierarchical brain networks, and modulate the influence of neural activity from lower-order upon higher-order regions (TRL1). The emerging data suggests that there may well be a useful role for these agents in the treatment for major depression, anxiety and drug addiction and in particular in some problematic circumstances where current treatment options are limited, prognosis is unfavourable, data suggests scant, if any, objective benefit of current therapy options and clinicians have historically adopted a distrustful attitude towards these difficult-to-treat patients.
Separating fervently held beliefs from objective facts is critical. This applies equally to the medical researchers conducting the genetic experiments and clinicians who treat diseases and conditions and to chemists who can assist by transitioning medical research data into practical pharmacological solutions.
In schizophrenia for instance, initially the genetic operating basis was dismissed because the assumptions about which genes were responsible was out by a country mile and the conceptual basis itself was far too simplistic to prompt a search in the right direction.
Further studies revealed that schizophrenia is not one disease but several genetically distinct ones (and hence the way drugs work will differ among these). It is now known that a total of 42 genetic clusters working in tandem are responsible for the symptoms of eight distinct disorders, all falling under one clinical label. So far, this new data has not seemingly affected how psychiatrists prescribe drugs or how they treat these patients.
Beyond the 5-HT receptor class, there are a myriad of potential targeting systems emerging from medical research results.
Neuroplasticity in adults is possible
Neuronal plasticity peaks early in life during critical periods and normally declines with age. Neuroscientists recently showed that it’s possible to return some of this plasticity to the brains of mice by tweaking a single gene (TRL1) (doi: 10.1073/pnas.1700866114). They targeted the expression of the Arc protein and genetically deleted its coding from DNA. This presents an enticing novel molecular target that could be used to fight age-related cognitive decline in humans. Moreover, the neuroscientists were able to restore the plasticity in the visual cortex of eyes of adult mice, potentially providing a gateway to developing means to restore sight, heal macular degeneration and correct age-related visual deficiencies. There are no effective treatment tools at present, and chemists are notable by their absence in this area.
The same result had serendipitously been obtained previously when neuroscientists (neuropsychiatrists) used a widely prescribed antidepressant drug called fluoxetine ‘off-label’ (TRL1) (doi: 10.1126/science.1150516). These researchers were able to restore the vision of adult rats while in the process of opening the door to unravelling new mechanisms for the therapeutic effects of antidepressants and for the pathophysiology of mood disorders. Alessandro Sale, the co-author on this major paper was also the editor and co-author of the first chapter of Environmental experience and plasticity of the developing brain (see review December 2017/January 2018, page 32.)
A significant number of similarly identified targets have been reported. For example, the gene Lsamp regulates emotional and social behaviour (TRL1) and could potentially be the gateway to cures in a range of psychiatric illnesses and drug dependency.
The potential targeting information available is at times very precise. For example, medical researchers are pinning down specific key molecules in the development and pathogenesis of emotion-related behaviour. One example is netrin-G1 which is a glycosyl-phosphatidylinositol-anchored synaptic adhesion molecule whose deficiency results in impaired fear-like and anxiety-like behaviours under specific circumstances. Genetic deletion of netrin-G1 in cortical excitatory neurons resulted in altered anxiety-like behaviour, but intact fear-like behaviour, whereas loss of netrin-G1 in inhibitory neurons resulted in attenuated fear-like behaviour, but intact anxiety-like behaviour.
From assumption to opportunity
We’re starting to unravel the genetics of pathology in mental health and degenerative diseases among a plethora of neural-wiring-related applications potentially amenable via targeting the receptors used in the command and control of these pathways. This is the point where a gulf exists between existing medical researchers and those who can transform their discoveries into practical solutions. Many of our most cherished and widely long-held assumptions are proving to be false. We’ve never seriously looked at neuroplasticity because we haven’t believed the possibilities.
‘Use it or lose it’ is the core operating principle of neuroplasticity. The same applies to these current and emerging golden opportunities.