Glimpses of chemistry in Australia
In 1917, Australia was still at war. The conflict that major protagonists had expected to be over by Christmas 1914 was dragging on. Prime Minister W.M. (Billy) Hughes, re-elected in May as the head of the new coalition Nationalist Party, was preparing a second conscription referendum. In December, it was defeated. Life went on, however, and the Trans-Continental Railway linking Melbourne and Fremantle was finally completed.
To all this we can add the formation of the Australian Chemical Institute as perhaps the greatest chemical event of the year. At that time, many Australian chemists were in Britain, toiling in the munitions industry, and perhaps some of their output was included in the 454 tonnes of ammonal (a combination of ammonium nitrate and aluminium powder) that was exploded under German lines at Messines Ridge in June.
Import replacement in Australia had begun with the establishment of a wood distillation plant near Melbourne, where methanol and acetic acid were produced from Eucalytpus regnans. Acetone, produced by dry distillation of calcium acetate, was then used in the production of cordite, a combination of nitroglycerine and nitrocellulose. Shmith and Nicholas, having worked out the process for the manufacture of acetylsalicylic acid, were marketing Aspro as a replacement for Bayer’s Aspirin.
The Commonwealth Advisory Council on Science and Industry (a forerunner of CSIR) had considered the need for the chemical industry to produce other chemicals formerly imported from enemy countries, with emphasis on potash for use in fertilisers and on industrial alcohol. Rain-spoiled wheat from Victoria and South Australia was suggested as a source of alcohol, and the Society of Chemical Industry of Victoria heard a lecture about alcohol as a future substitute fuel.
It could not have been evident at the time, but major contributions to chemistry were to be made by three Australians born in 1917, all of whom later graduated from the University of Sydney. John Cornforth (1917–2013), an organic chemist, completed his doctorate at Oxford in 1941 and pursued his career in Britain. In 1977, he shared the Nobel Prize with Vladimir Prelog for their studies of the stereochemistry of enzyme actions. Ronald Nyholm (1917–71), an inorganic chemist, already had substantial research experience before he went to University College London (UCL) for PhD studies. After a couple of years at the University of New South Wales, he returned to UCL. Ernest Ritchie (1917–76) remained at the University of Sydney where he earned the DSc for his research on Australian plant products
Possibly Australia’s first school chemistry book was published in 1917. It was Australasian text book of chemistry. Part 1, written by W.R. Jamieson, the senior chemistry master at Scotch College, Melbourne. Known to his pupils as ‘Tort’ (for ‘retort’), Jamieson wrote that his book ‘covers a two-years’ course for boys (and girls, too, for that matter) from the age of 14 to that of 16’.
Glimpses of the year’s research
For research in chemistry, I turned to the Annual Reports on the Progress of Chemistry published by the Chemical Society of London. This series began in 1904 with the aim of summarising and commenting on work published during the calendar year. The things that caught my eye were mostly those that were to become icons of chemistry during the next century.
The 1917 reviews included reports of shortages of material such as glycerol (no doubt for the manufacture of glyceryl trinitrate aka nitroglycerine), platinum (leading to the use of gold and palladium in chemical apparatus) and microbalances of European make. Horse chestnuts and acorns were reportedly being used for food in Germany. Although the Zeitschrift für physikalische Chemie had ceased publication because suitable paper was unavailable, other journals were received from Germany and the publications were referenced as often as those from Britain and America.
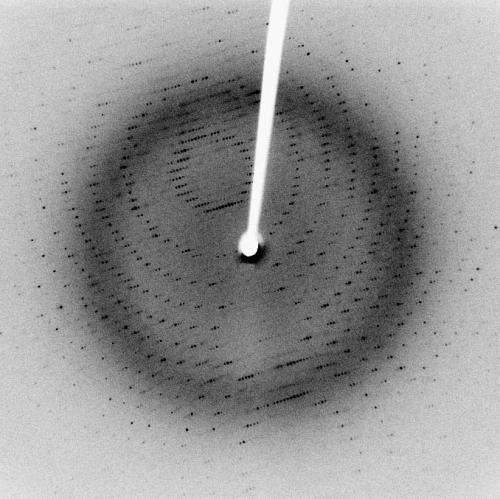
A good deal of space in the General and Physical Chemistry section was devoted to X-ray crystallography and there was a Crystallography and Mineralogy section as well. The highlight was the introduction by P. Debye and P. Scherrer of a method for recording data for crystals of ‘microscopic or submicroscopic dimensions’. The crystalline powder was placed on the axis of the cylindrically arranged film and irradiated with X-rays. Debye and Scherrer used this technique to study graphite and amorphous carbon and to show that they have the same (trigonal) structure. I remember using their method, half a century later, in a third-year experiment in which the X-ray pattern was used to show that a spinel had been formed from two oxides that I had sintered together. A little petroleum jelly was used to stick the powdered product to a glass fibre that was mounted in the X-ray beam.
At about this time, the Russian crystallographer Evgraf Stepanovich Fedorov (1853–1919) was publishing criticism of the Braggs’ work, but inability to read Russian meant that the reviewer, H.M. Dawson, was unable to assess Fedorov’s contentions. The Braggs had been awarded the Nobel Prize in Physics for 1915 but X-ray crystallography was still very new. A mini-(literally)-highlight was the report of its use by a former student of Federov, T.V. Barker at the University of Oxford, in his examination of a ‘minute crystal’ of ‘intestinal’ origin that was provided by a Belgian medical researcher. Consulting a table of unit cell dimensions published by Federov, Barker thought the 229-milligram crystal was either potassium niobate, methyl di-isopropyl sulfine hexachloroplatinate or phenyl salicylate (Salol), and the last of these was confirmed by measuring the melting point (43ºC) of a ‘tiny splinter’ (<0.1 milligram) of the crystal. Why was it in the intestine? Well, it’s not very soluble in water and was in use as an analgesic and anti-rheumatic, but Barker didn’t say.
The X-ray boom had re-opened older questions of structure, such as whether there were molecules in crystalline inorganic solids. X-ray methods were showing atomic lattices but the molecule concept was hard to put aside and one researcher quipped that rock salt could just as easily be regarded as consisting of [NaCl6] and [ClNa6], but the reviewer observed that ‘other practicing X-ray analysts leave such questions severely alone’.
The reviewer of Inorganic Chemistry, E.C.C. Bayly, felt that he was going beyond chemistry in covering the physical phenomenon of the phosphorescence of zinc sulfide, which was widely thought to be due to the presence of various impurities. A Glasgow research group led by A.W. Stewart (before he started writing detective stories as ‘J.J. Connington’) had reported that pure zinc sulfide does phosphoresce but only if it has been heated for long periods at 650–900°C to induce the formation of a semicrystalline state. Other researchers had demonstrated that the phenomenon could only be observed in this and other solids when impurities like manganese were present, and by mentioning their work, I think Bayly was begging the question ‘Was Stewart’s material really pure?’.
In the Organic Chemistry section, the synthesis by German chemists of lead tetra-alkyls containing four primary alkyl groups caught my eye. Starting with tetramethyl lead, a methyl group was removed by reaction with halogen at low temperature, and this substance was treated with a Grignard reagent to attach a new alkyl group. Two more halogenations, each removing a methyl group, and successive use of other Grignards, gave methylethyl-n-propyl-n-butyl lead.
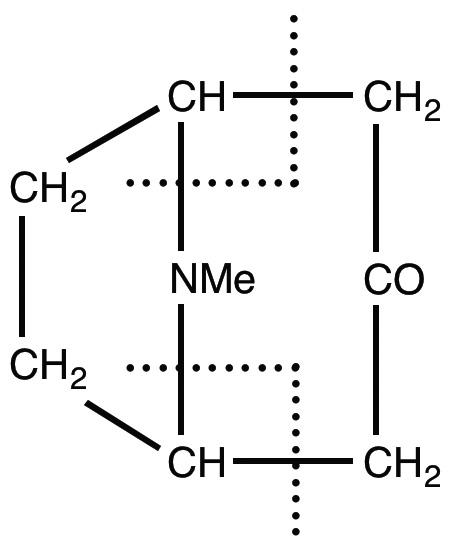
There was, of course, a fair bit of natural product chemistry. From an aqueous extract of the fruit of the ‘avocado pear’ (Persea gratissima), American researchers isolated a seven-carbon sugar, a mannoketoheptose. Its reduction product, a heptahydroxy alcohol, had been isolated from the same source by Emil Fischer in 1890. My award for the best paper of 1917 goes to Robert Robinson, who published his biomimetic synthesis of the alkaloid tropinone, and generalised the concept of phytochemical syntheses by suggesting how a series of simple reactions could effect the syntheses of a range of alkaloids. ‘Without exaggeration’, the reviewers said, ‘it may be said that this paper marks an epoch in the consideration of alkaloid syntheses’. Robinson had only recently returned to England to take up the chair in Liverpool after three years in Sydney as foundation professor of organic chemistry there.
Reviewing Analytical Chemistry, C. Ainsworth Mitchell wrote that ‘the influence of the war on chemistry in general is reflected in the utilitarian character of many of the recent contributions to analytical chemistry’, dealing with ‘industrial by-products or substances to take the place of those no longer available’. For example, with the best micro-analytical balances unobtainable, an ordinary balance had to be used and the quantity of substance necessary for analysis had to be increased to 11–22 milligrams. A tube packed with soda-lime was used in place of the potash absorption bulb for capture of carbon dioxide.
In the section on Agricultural Chemistry and Vegetable Physiology, E.J. Russell reported that ‘enormous quantities of ammonia and of nitric acid are now being synthesized on the Continent’ but went on to write about British work with calcium cyanamide. He did refer to a publication in the Journal of the Society of Chemical Industry in which the role of Haber was made explicit. I revert to Australia for a minute: the professor of chemistry at the University of Adelaide, Edward Rennie, in his Commemoration Address in December, described the development of chemistry in connection with the war, and referred coyly to ‘a German chemist’ who had mastered the art of making ammonia from atmospheric nitrogen. Like so many commentators before him, Rennie deplored the lack of support for science in Britain. A reporter for the Advertiser paraphrased him thus: while other countries were treating science as a sort of hobby, Germany was exploiting it for purposes, not merely with industry, alas, but of war’. Rennie went on to describe German chemistry as it applied to munitions, food, poisonous gases, manures, dyes and drugs. Was he a little bit envious of German progress?
Some work of the Australian biochemist Thorburn Brailsford Robertson (1884–1930) was mentioned in the section on Physiological Chemistry compiled by Frederick Gowland Hopkins. Robertson completed his BSc at the University of Adelaide and PhD at the University of California where he was appointed full professor in 1917. After a couple of years in Toronto, he returned to Adelaide but then in 1927 was appointed by CSIR to form their Division of Animal Nutrition. In 1917, he reported the isolation from the pituitary gland of a substance he called tethelin that promoted growth and healing. The molecule was said to contain phosphorus and nitrogen atoms in the ratio 4:1, yielded i-inositol on hydrolysis, and gave a colour test for an imidazole group. Despite later investigations by Australian biochemists, its nature remains a mystery, although there has been extensive research on growth factors.
The 1917 prize for whimsy goes to B. Bloch who identified a black pigment formed by oxidation of 3,4-dihydroxyphenylalanine, to which he gave the name ‘dopa’. It wasn’t dopey at all, and despite Gowland’s deprecation of it as an ‘uneuphonious portmanteau name’ that could lead to ‘dopaoxydase’ for the enzyme, and ‘dopamelanin’ for the black pigment, the shorthand ‘dopa’ is widely used today for the amino acid.
But no Nobel
Given all the wonderful chemistry of the previous decade, I was surprised to find that the Nobel Prize in Chemistry was not awarded in 1917 (it wasn’t in 1916, either). Among the names of the 11 unsuccessful nominees in 1917 were some recognisable ones like those of Walther Nernst and Fritz Pregl (Nobel laureates in 1920 and 1923, respectively) and Theodor Curtius, who never got a gong. It’s possible that the selectors were wary of awarding the Prize to a German chemist but there were worthy nominees, I think, from other countries. Fritz Haber wrote later that ‘I have no doubt that the political situation makes it impossible for a German nominated by Germans to be considered in Stockholm’ but nonetheless, he was awarded the Nobel Prize in 1918. Not for gas warfare, of course, but for the ammonia synthesis, for which he was nominated by German engineer Ludwig Prandtl.